Recent Posts
Uninstall and Remove Voicemeeter, Step by Step Virtual Mixing Console – 2023 Guide
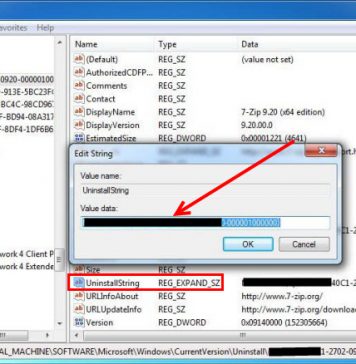
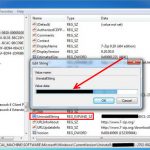
Do you need to remove the Voicemeeter, the virtual mixing console, to solve some problems? Are you looking for an effective solution to completely...
Upstart.com / MyOffer Reviews and Offer Code (Do you want to use it?) –...
Assessment of personal loans
If you're lucky enough to get an UpStart loan for U.S. mail advertising, you should have a good credit rating. Congratulations!
The...
Visit GetInkPreferred.com Invitation Number – Apply Online (Ultimate 2023 Guide and Review)
Receive your favourite ink offer
Request a Chase Ink credit card with a 12-digit invitation number.
This mail offer is aimed at pre-selected consumers.
Go to www.getinkpreferred.com...
Walmart Card Offer: Pre-screen Credit Card-Get 3 Percent Cash Back – 2023 Guide
Walmart.com Card Offer /Credit Card Overview
Applicants must provide a pre-approved approval number and a surname.
A Walmart credit card is a YES PLUS...
Watch Game Of Thrones Season 7 Full Episodes Online – 2023 Guide
If you want to view the entire Throne 7 season online and even download the full episodes without signing up or paying a penny,...