Recent Posts
Uninstall and Remove Eurodict Spanish Dictionary 3.1 Step by Step 2023 Guide
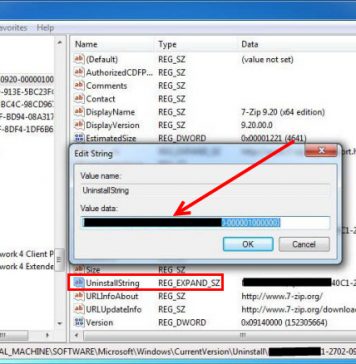
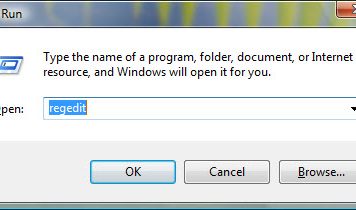
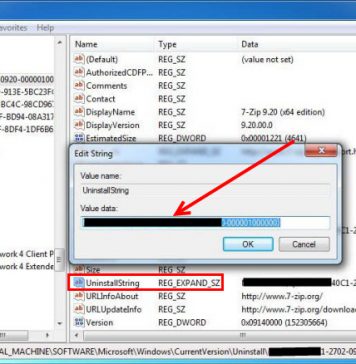
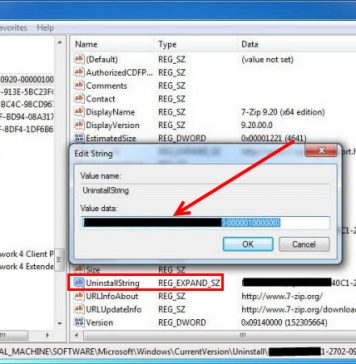
Do you need to delete Eurodict Spanish Dictionary 3.1 to fix some problems? Are you looking for an effective solution to completely delete all...
Uninstall and Remove Guild Wars 2 Step by Step 2023 Guide
Do you need to remove Guild Wars 2 to solve some problems? Are you looking for an effective solution to completely delete all files...
Uninstall and Remove Infineon TPM Professional Package Step by Step – 2023 Guide
Do you need to uninstall the Infineon TPM Professional software package to solve some problems? Are you looking for an effective solution to completely...
Uninstall and Remove Intel Rapid Technology Step by Step – 2023 Guide
Do you need to uninstall Intel Rapid Storage Technology to solve some problems? Are you looking for an effective solution to completely delete all...
What Does That Mean When a Girl is Calling You a Hun
If you recently received a phone call from a young girl, you probably wonder why and what it has to do with the way...