Recent Posts
How to Perform Regular Maintenance on Your Heavy-Duty Vehicles
Nina Smith - 0
Heavy-duty vehicles like trucks, buses, and construction equipment require regular maintenance, even though they're built to endure tough conditions. These machines are indeed resilient,...
How to Go Back to Work After Deep Vein Thrombosis – 2023 Guide
Deep Vein Thrombosis or DVT is one of the health diseases many people around the world experience. Unfortunately, it is not one of the...
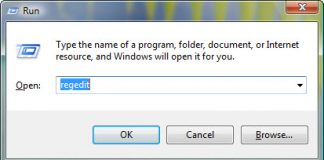
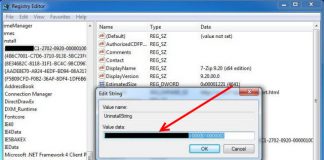
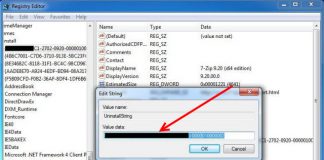
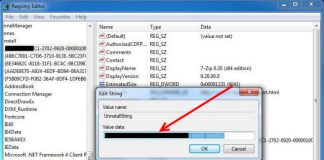
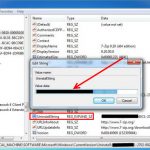
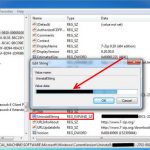
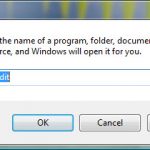
Uninstall and Remove iSEEK AnswerWorks English Step by Step – 2023 Guide
Do you need to remove the English runtime from iSEEK AnswerWorks to solve some problems? Are you looking for an effective solution to completely...
Uninstall and Remove the Legends League Step by Step – 2023 Guide
You have to remove League of Legends to solve some problems? Are you looking for an effective solution to completely delete all files from...
Uninstall and Delete Nexus Mod Manager Step by Step – 2023 Guide
Do you need to uninstall Nexus Mod Manager to fix some issues? Are you looking for an effective solution to completely delete all files...