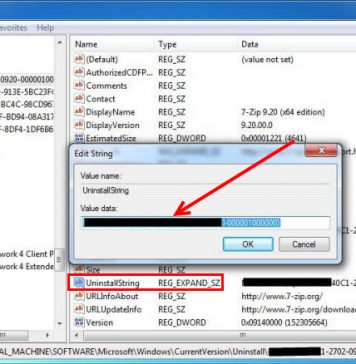
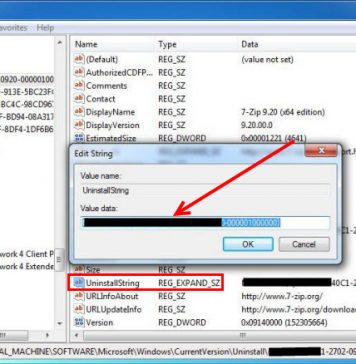
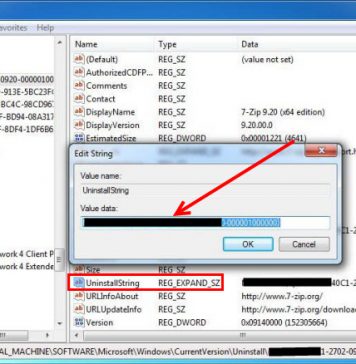
Recent Posts
Uninstall and Remove Quickset64 Step – 2023 Guide
Do you need to uninstall Quickset64 to fix some issues? Are you looking for an effective solution to completely delete all files from your...
Uninstall and Remove the Sky Go Player Step – 2023 Guide
Do you need to uninstall the Sky Go Player to fix some issues? Are you looking for an effective solution to completely delete all...
Uninstall and Remove VBCABLE-B, Virtual Audio Cable Step – 2023 Guide
Do you need to remove the VBCABLE-B, virtual audio cable, to solve some problems? Are you looking for an effective solution to completely delete...
Uninstall and Remove Voicemeeter, Step by Step Virtual Mixing Console – 2023 Guide
Do you need to remove the Voicemeeter, the virtual mixing console, to solve some problems? Are you looking for an effective solution to completely...
Upstart.com / MyOffer Reviews and Offer Code (Do you want to use it?) –...
Assessment of personal loans
If you're lucky enough to get an UpStart loan for U.S. mail advertising, you should have a good credit rating. Congratulations!
The...